What is Surface Tension??

'In physics, surface tension is a force present
within the surface layer of a liquid that causes
the layer to behave as an elastic sheet.
It is the force that supports insects that walk on water,
for example.' A quote from http://encyclopedia.kids.net.au/page/su/Surface_tension
within the surface layer of a liquid that causes
the layer to behave as an elastic sheet.
It is the force that supports insects that walk on water,
for example.' A quote from http://encyclopedia.kids.net.au/page/su/Surface_tension
How Does it Work??
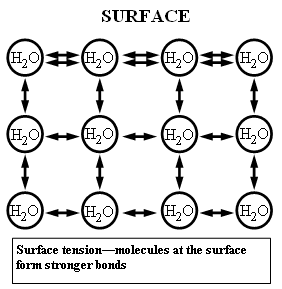
Surface tension is caused by the attraction between the hydrogen molecules of the liquid (hydrogen bonding). On the surface of the liquid, each particle is pulled in all directions by the other molecules. The molecules that are below the surface keep the ones on top of the water in place (by pulling the molecules on the top towards them) . This works because the outward attraction by the hydrogen molecules in the air, which is much less dense than the water, doesn't have any effect on the surface tension because of the density.
In this diagram, the water molecules at the top have stronger bonds with each other than the other particles, this causes the elastic-like sheet to form on the surface.